As many industries begin to move away from simple container packaging and shift to smart packaging, the potential for convenience and clarity increases. Unfortunately, with some of those advanced technologies comes an increased risk. For the pharmaceutical industry, this can mean having to contend with counterfeiting and the lack of information and visibility from raw material to consumer delivery (supply chain opacity).
It is not just a matter of risk, especially when weighing up the benefits they offer. By understanding the mechanics of some of the most prominent smart packaging for healthcare products, such as RFID, NFC and time-temperature indicators, the industry has the potential to turn traditional static labels into intelligent tools that increase compliance and patient safety.
With all of this in mind, how can smart packaging work for healthcare products to provide the right combination of clarity, compliance and safety? This guide by CCL Healthcare explains everything you’ll need to know about smart packaging and how it can be used in healthcare.
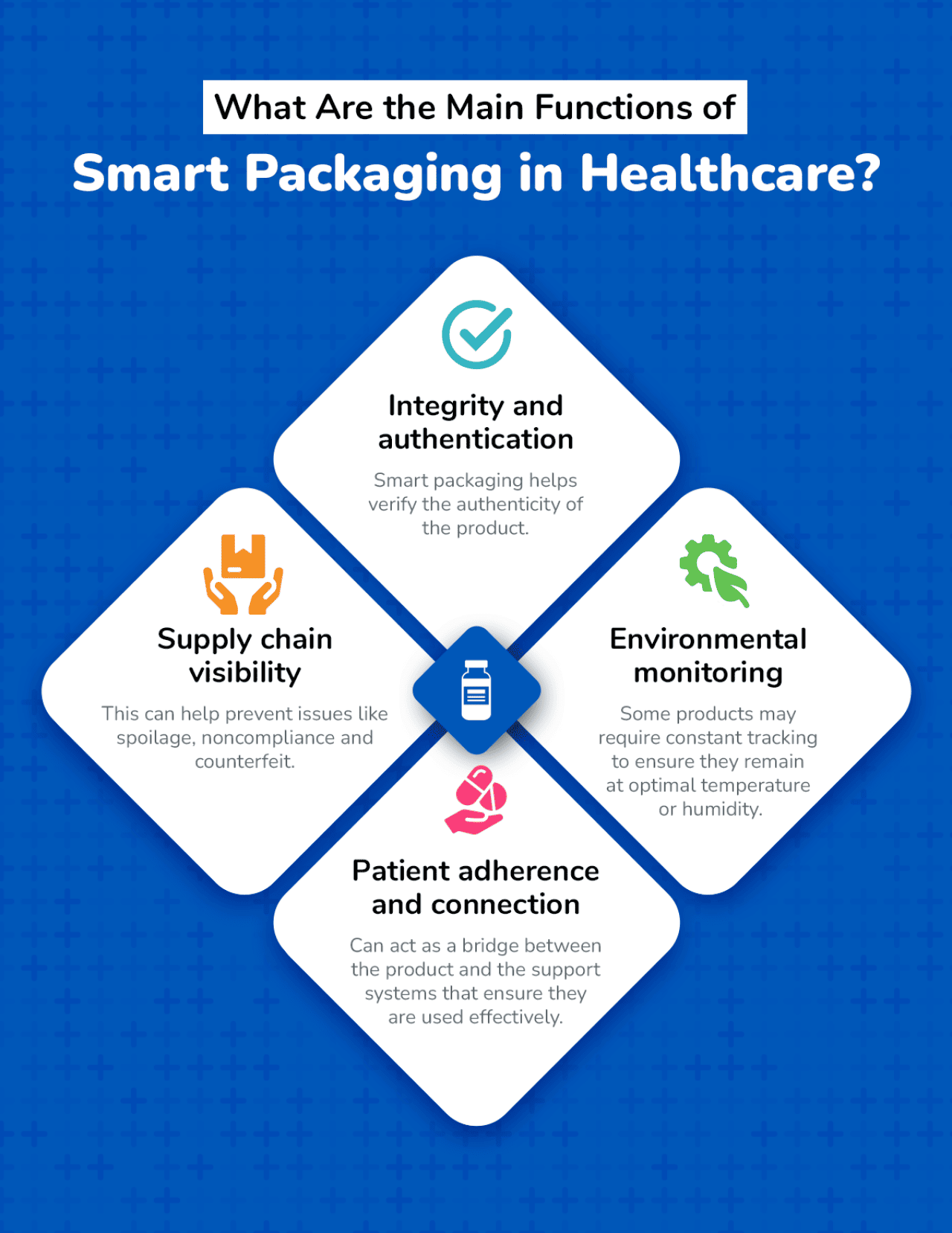
Smart packaging for healthcare products can provide many important functions for organizations, manufacturers and patients alike. With the smart medication packaging market expected to reach $57.52 billion by 2032, these functions are on track to continue growing significantly.
Some of the most essential main functions of smart packaging to be aware of in the healthcare industry include:
Understanding the main functions of smart packaging in healthcare is important. However, knowing the mechanics of how they work can be the key to determining which works well within your organizational goals.
RFID technology and NFC are two innovations that play important roles in fast-tracking and maintaining item serialization, authenticity verification, and supply chain visibility. Having access to the increased traceability of your healthcare products can ensure compliance with the Drug Supply Chain Security Act (DSCSA) in the United States, and the European Commission’s Falsified Medicines Directive (FMD).
While the specific connectivity and regulatory needs of an organization will depend on where in the world you operate, having increased awareness of how RFID and NFC work is the first step toward knowing how they can benefit you and your customers.
Radio-frequency identification is a form of innovative wireless technology. It consists of the RFID tag, or transponder, which contains a microchip and an antenna that can transmit or receive signals. The other part of an RFID is the reader, or interrogator, which releases radio waves to power the tags that provide essential data for tracking purposes.
RFID technology can come in both passive and active tags:
When the reader sends a signal to the antenna, whether passive or active, a chip will transmit the unique product data back to the reader, which can be fed into a computer system. This process can help manage assets and capture information with ease.
NFC is a form of wireless technology that allows devices within a short range (usually under 4 centimeters) to exchange data. On items like drug packets and containers, medical and pharmaceutical staff or consumers can confirm authenticity and track movements by using smartphones.
While using NFC in healthcare packaging can support drug authentication, it can also contribute to supporting patients via wearable sensors (helping monitor blood pressure and heart rates), patient tracking, and records and diagnostics sharing.
The simplicity of NFC tags and their low power usage makes them ideal for secure data flow between devices and fast access to vital information like vaccination records.
Time-temperature indicators act as a measuring stick for the amount of degradation a drug has been subjected to. It could be considered similar to how a thermometer works, but a TTI actually measures the cumulative heat of a drug.
Some of the key benefits of a TTI for organizations and patient well-being include:
Time-temperature indicators give off a visual (or sometimes digital) signature through chemical, physical or biological changes that react to cumulative temperature exposure. This design is similar to a visual trail that provides details on product results and color changes when drugs exceed temperature limits over time.
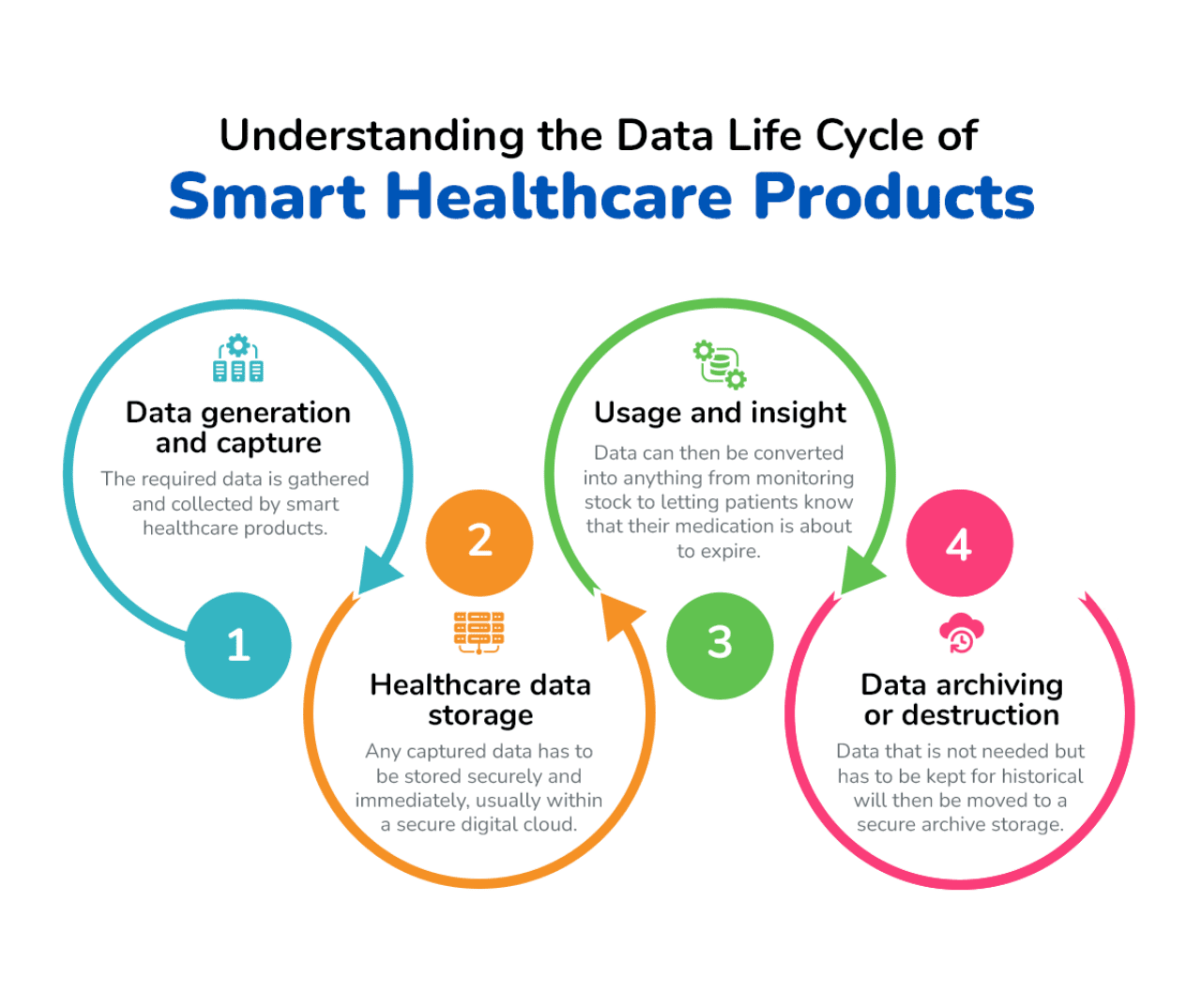
Smart packaging for healthcare products involves many different factors and stages as part of its life cycle. The data involved in this process is perhaps the most important element. From the generation of data from connected devices right up to the destruction of data that’s no longer needed, it is important for patients and organizations to know where that vital data goes and how well it’s protected.
While there are several smart healthcare packaging trends that can benefit organizations industrywide, they all typically involve the following data life cycle journey:
It’s important to remember that all of these steps in the data life cycle of smart healthcare products, including packaging, requires authorization and adherence to use data sensibly and respectfully.
Smart packaging plays a pivotal role in the constantly evolving pharmaceutical industry. As modern consumers expect faster, more efficient methods of product delivery and information, smart packaging offers a multitude of benefits for patients, manufacturers and leading pharmaceutical organizations.
The global pharmaceutical industry has continued to grow over the last two decades, reaching $1.7 trillion. As smart packaging for healthcare products continues to evolve and generate income, everyone can enjoy the benefits of its innovative advancements, including:
Smart packaging technologies are redefining how several industries across the world operate, and pharmaceuticals are certainly no exception. With artificial intelligence-related drug developments, telemedicine and other advancements helping connect products with the digital market, it’s becoming more common for physical containers to be useful active parts of the product life cycle.
Like any evolving technology, a gap is beginning to emerge between those who benefit from smart packaging and those who don’t. Industries, processes and patients risk being left behind and missing out on these innovations if they aren’t fully embraced across the board.
Every industry can benefit from accurate inventory tracking and the ability to keep on top of the environmental conditions of their goods. In the case of pharmaceuticals, having constant access to information like product health and temperature updates can help reduce the amount of waste created from expired or damaged healthcare products.
This focus on reducing waste can also lead to a significant decrease in operational costs, while also having insights into clearer demand forecasting by making use of the real-time data available.
Planning for any potential problems in healthcare products can be tedious and complex. However, it is still an essential part of being prepared for any “worst case scenario” situations that can affect efficiency or safety. Healthcare companies are under pressure to react quickly in the event of emergency product recalls to prevent faulty or dangerous medication from reaching patients.
Being able to track down each individual product is an effective risk management tool, especially in situations with potential financial liability. In many cases, smart packaging can provide an almost instant update on a potential risk.
How is smart packaging transforming the pharmaceutical industry for integrity across the board? With the delicate nature of pharmaceutical regulations, companies have strict requirements to follow to remain compliant. A clearly defined digital identity for every healthcare packaging item creates a trail and record that’s impossible to deny and easy to locate.
Having supply chain visibility of this magnitude provides ultimate product integrity for smart and intelligent packaging, from manufacturing to the very end of the product’s journey. This can help protect patients while also maintaining the organization’s brand reputation and integrity.
Smart packaging technologies in healthcare offer a roadmap that converges safety, compliance and efficiency, helping improve the lives of patients and life cycles of products. Pharmaceutical organizations can benefit from implementing innovative technology and embracing the shift from past practices to future outlooks.
For pharmaceutical manufacturers and key decision-makers, taking the time to assess product lines to check for smart packaging capacity can be the difference between a strong and efficient supply chain and being left behind by a constantly evolving industry.
The ability to place the latest intelligence directly into a medical product container can be the first step to discovering unlimited potential data insights. That data can define an exciting new future that streamlines key data while protecting sensitive information, which elevates the entire experience for manufacturers, organizations and patients in need of quality healthcare.
This story was produced by CCL Healthcare and reviewed and distributed by Stacker.
Reader Comments(0)